글로벌 연구동향
분자영상 및 방사화학
- [J Org Chem .] Direct 18F-Fluorosulfurylation of Phenols and Amines Using an [18F]FSO2+ Transfer Agent Generated In Situ
[18F]FSO2+ 이동제를 사용한 페놀 및 아민의 직접 18F- Fluorosulfurylation연세대, UNIST / 김민평, 전중현*, 홍성유*
- 출처
- J Org Chem .
- 등재일
- 2023 May 5
- 저널이슈번호
- 88(9):6263-6273.
- 내용
-
Abstract
We report the direct radiofluorosulfurylation method for the synthesis of 18F-labeled fluorosulfuryl derivatives from phenols and amines using an [18F]FSO2+ transfer agent generated in situ. Nucleophilic radiofluorination is achieved even in a hydrous organic medium, obviating the need for azeotropic drying and the use of cryptands. This unprecedented, operationally simple isotopic functionalization facilitates the reliable production of potential radiotracers for positron emission tomography, rendering facile access to SuFEx radiochemistry.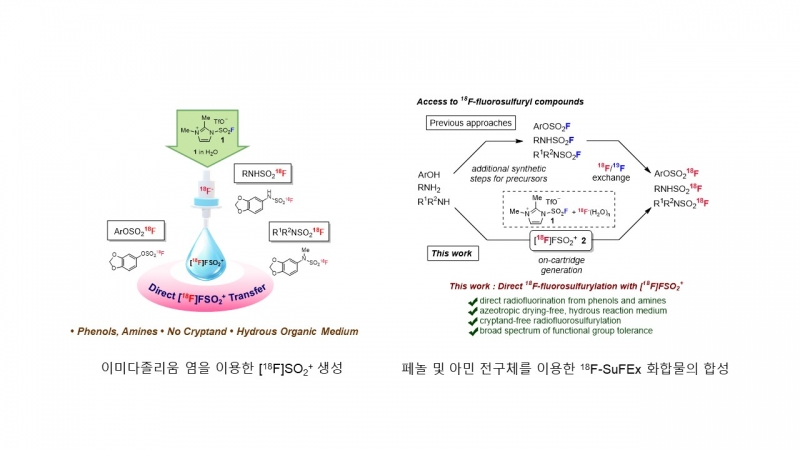
Affiliations
Min Pyeong Kim 1, Hojin Cho 2, Swatilekha Kayal 1, Min Ho Jeon 1, Jeong Kon Seo 3, Jeongmin Son 2, Jinsil Jeong 2, Sung You Hong 1, Joong-Hyun Chun 2
1Department of Chemistry and Department of Chemical Engineering, Graduate School of Carbon Neutrality, Ulsan National Institute of Science and Technology (UNIST), Ulsan 44919, Republic of Korea.
2Department of Nuclear Medicine, Yonsei University College of Medicine, Seoul 03722, Republic of Korea.
3UNIST Central Research Facility, Ulsan National Institute of Science and Technology (UNIST), Ulsan 44919, Republic of Korea.
- 연구소개
- 본 논문은 최근 들어 유기화학 분야에서 큰 관심을 받고 있는 SuFEx (sulfur-fluoride exchange reaction) 화학의 SuFExable 분자에 불소-18을 도입하는 연구 결과를 소개한 것입니다. 특히, SuFEx chemistry에 사용되는 아릴 플루오로 설페이트(aryl fluorosulfate)와 설파모일 플루라이드(sulfamoyl fluoride)계열의 화합물에 손쉽게 불소-18을 도입함으로써, 향후 황-불소(sulfur-fluorine) 결합을 가지고 있는 다양한 분자에 동위원소를 도입해 방사성의약품으로 사용할 수 있는 길을 열어 놓았습니다. 진단 및 치료용 방사성의약품의 화학적인 구조가 점점 다양해져 가고, 이러한 화학 구조에 효율적으로 동위원소를 도입할 수 있는 표지방법의 연구는, 복잡한 화학 구조를 가진 테라노스틱 방사성의약품을 성공적으로 개발하는 데 꼭 필요한 기술입니다. 본 논문에서는 QMA에 흡착된 불소-18을 이미다졸 설포닐 염(imidazolium sulfuryl fluoride)을 사용하여 용출한 뒤, 페놀 또는 아민 계열의 화합물에 직접 –SO218F를 도입할 수 있음을 소개하고 있습니다.
- 덧글달기
- 이전글 [Mol Pharm .] Theranostic Surrogacy of [123I]NaI for Differentiated Thyroid Cancer Radionuclide Therapy
- 다음글 [Biochem Biophys Res Commun.] Evaluation of PSMA target diagnostic PET tracers for therapeutic monitoring of [177Lu]ludotadipep of prostate cancer: Screening of PSMA target efficiency and biodistribution using [18F]DCFPyL and [68Ga]PSMA-11