글로벌 연구동향
분자영상 및 방사화학
- [J Immunother Cancer] Relaxin-expressing oncolytic adenovirus induces remodeling of physical and immunological aspects of cold tumor to potentiate PD-1 blockade 방사면역치료및 면역관문억제제 치료 효과 향상을 위한 유전자병용치료법 개발
KIRAMS, 한양대 / 정보경, 고혜영, 김진수*, 윤채옥*
- 출처
- J Immunother Cancer
- 등재일
- 2020 Aug
- 저널이슈번호
- 8(2):e000763. doi: 10.1136/jitc-2020-000763.
- 내용
-
Abstract
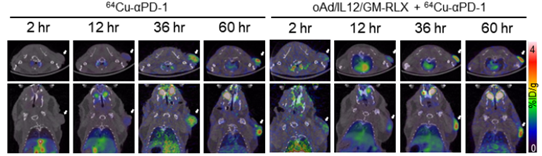
Background: Currently, several antibody (Ab)-based therapies have shown excellent therapeutic effects in the clinic. Nonetheless, Ab penetration into tumor tissues is limited due to abnormal vasculature, tumor interstitial pressure, and excessive extracellular matrix (ECM) accumulation, thus demanding novel strategies to overcome these barriers.Methods: The intratumoral distribution of therapeutic Abs were detected by fluorescence microscopy or positron emission tomography in both human gastric xenograft and syngeneic pancreatic hamster tumor models. The antitumor efficacy by combination of oncolytic adenovirus (Ad), which coexpresses relaxin (RLX), interleukin (IL)-12, and granulocyte macrophage colony-stimulating factor (GM-CSF) (oAd/IL12/GM-RLX) and antibody against the programmed cell death protein 1 (αPD-1) was examined in hamster subcutaneous and orthotopic pancreatic tumor models. The immunological aspects of these combination therapy regimen were assessed by flow cytometry or immunohistochemistry in subcutaneous hamster tumor models.
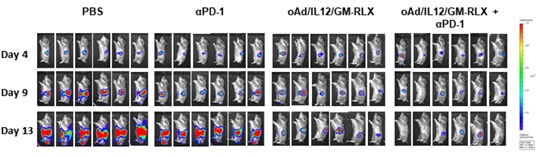
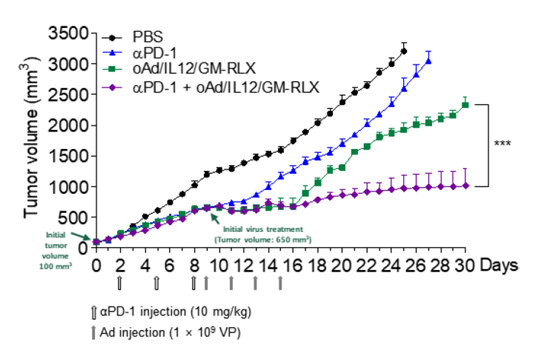
Results: Relaxin-expressing oncolytic Ad effectively degraded tumor ECM and enhanced the tumor penetration of trastuzumab in comparison with trastuzumab monotherapy. Based on these results, an oAd/IL12/GM-RLX was used to enhance the potency of immune checkpoint blockade. The combination of the oAd/IL12/GM-RLX and αPD-1 promoted a concomitant degradation of the tumor ECM and amelioration of the immunosuppressive tumor niches, ultimately enhanced intratumoral infiltration of both αPD-1 and activated T cells. Of note, the combination therapy was able to elicit a potent and durable antitumor immune response against cold tumors that were refractory to immune checkpoint inhibitor monotherapy.
Conclusions: Our findings are the first to demonstrate that expression of four genes (IL-12p35, IL-12p40, GM-CSF, and RLX) mediated by a single oncolytic Ad vector can promote remodeling of both physical and immunological aspects of the tumor niches to overcome the major limitations of Ab-based therapies that have emerged in recent clinical trials.
방사성동위원소인 Cu-64를 표지한 면역관문억제제의 치료 효과를 증진시키기 위하여 면역억제환경 및 세포외 기질 등의 항종양 치료 효과 저해요인을 극복할 수 있는 유전자치료제를 병용투여하여 치료 효과가 증진됨을 PET 및 형광영상장비를 통하여 확인하고 종양크기를 측정하여 입증하였습니다.
Affiliations
Bo-Kyeong Jung # 1 , Hae Young Ko # 2 , Hyunji Kang 2 , JinWoo Hong 1 3 , Hyo Min Ahn 1 3 , Youjin Na 1 , Hyeongi Kim 2 , Jin Su Kim # 4 5 , Chae-Ok Yun # 6 3 7
1 Department of Bioengineering, College of Engineering, Hanyang University, Seoul, Korea (the Republic of).
2 Division of RI Application, Korea Institute of Radiological and Medical Sciences, Seoul, Korea (the Republic of).
3 Department of Research and Development, GeneMedicine Co., Ltd, Seoul, Korea (the Republic of).
4 Division of RI Application, Korea Institute of Radiological and Medical Sciences, Seoul, Korea (the Republic of) chaeok@hanyang.ac.kr kjs@kirams.re.kr.
5 Radiological and Medico-Oncological Sciences, University of science and technology (UST), Seoul, Korea (the Republic of).
6 Department of Bioengineering, College of Engineering, Hanyang University, Seoul, Korea (the Republic of) chaeok@hanyang.ac.kr kjs@kirams.re.kr.
7 Institute of Nano Science and Technology (INST), Hanyang University, Seoul, Korea (the Republic of).
# Contributed equally.
- 키워드
- immunotherapy; lymphocytes, tumor-infiltrating; oncolytic virotherapy; radioimmunotherapy; tumor microenvironment.
- 연구소개
- 방사면역치료 및 최근 각광받는 면역관문억제제 치료 효과 향상을 위하여 위암 및 췌장암 등의 고형암에서 세포외 기질을 분해하고, 면역억제환경을 증진 기능이 있는 유전자치료 활용할 때 기존 항체 치료의 한계를 극복할 수 있음을 입증하였습니다.
- 덧글달기