글로벌 연구동향
방사선생물학
- [Mol Cells.] Radiation-Induced CXCL12 Upregulation via Histone Modification at the Promoter in the Tumor Microenvironment of Hepatocellular Carcinoma.
아주대 / 안학준, 이종수*
- 출처
- Mol Cells.
- 등재일
- 2019 Jul 31
- 저널이슈번호
- 42(7):530-545. doi: 10.14348/molcells.2019.2280.
- 내용
Abstract
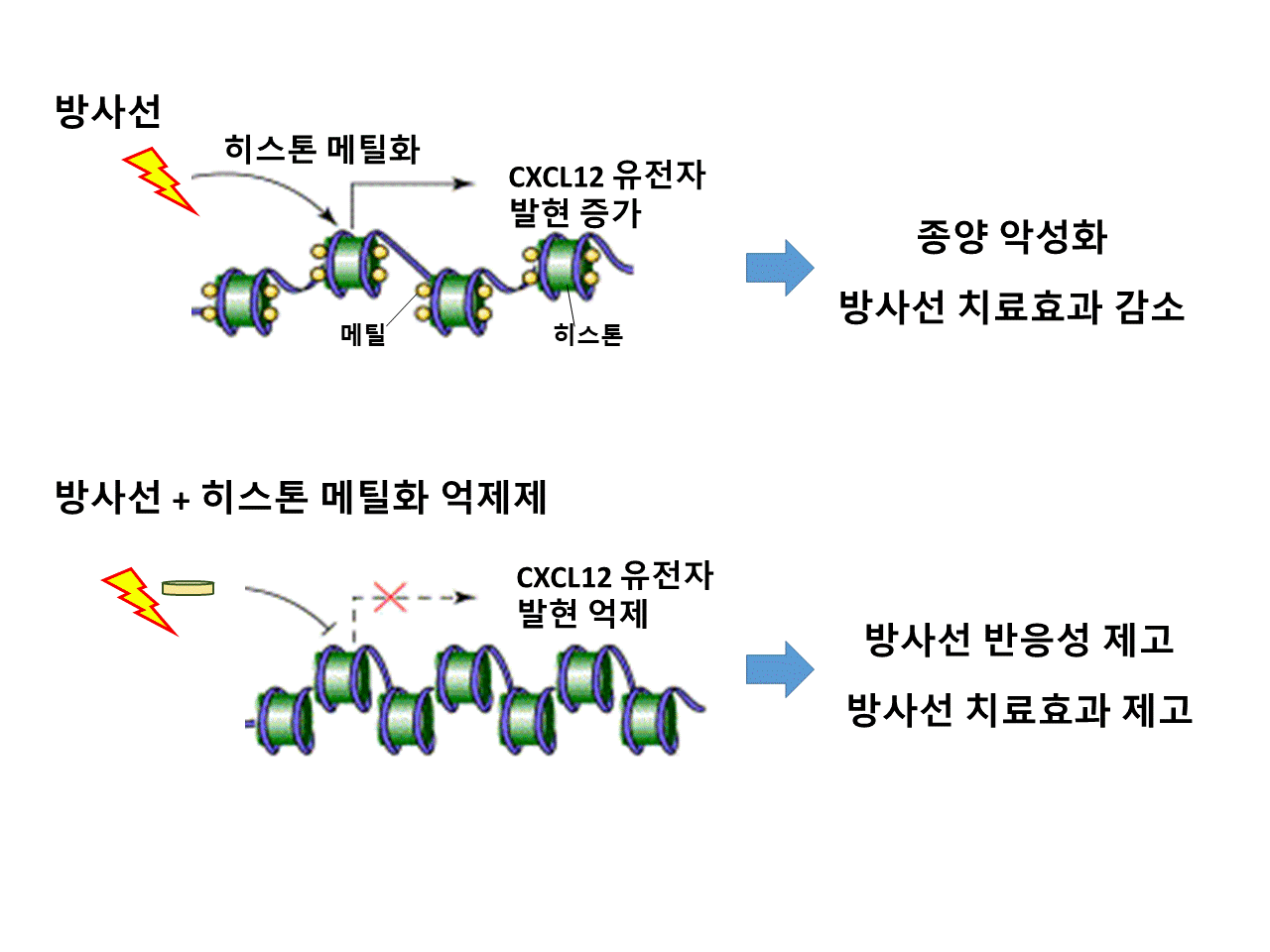
Tumor cells can vary epigenetically during ionizing irradiation (IR) treatment. These epigenetic variegations can influence IR response and shape tumor aggressiveness. However, epigenetic disturbance of histones after IR, implicating in IR responsiveness, has been elusive. Here, we investigate whether altered histone modification after IR can influence radiation responsiveness. The oncogenic CXCL12 mRNA and protein were more highly expressed in residual cancer cells from a hepatoma heterotopic murine tumor microenvironment and coculture of human hepatoma Huh7 and normal IMR90 cells after radiation. H3K4 methylation was also enriched and H3K9 methylation was decreased at its promoter region. Accordingly, invasiveness and the subpopulation of aggressive CD133+/CD24- cells increased after IR. Histone demethylase inhibitor IOX1 attenuated CXCL12 expression and the malignant subpopulation, suggesting that responses to IR can be partially mediated via histone modifications. Taken together, radiation-induced histone alterations at the CXCL12 promoter in hepatoma cells are linked to CXCL12 upregulation and increased aggressiveness in the tumor microenvironment.
Author informationAhn HJ1,2, Hwang SY1,3,2, Nguyen NH1, Lee IJ4, Lee EJ4, Seong J4, Lee JS1,3.
1
Department of Life Science, College of Natural Sciences.
2
These authors contributed equally to this work.
3
Functional Cellunomics Institute, Ajou University, Suwon 16499, Korea.
4
Department of Radiation Oncology, Yonsei University Medical College, Yonsei University Health System, Seoul 03722, Korea.
- 키워드
- CXCL12; histone modification; malignancy; radiation; tumor microenvironment
- 연구소개
- 방사선 치료 효과를 제고하기 위하여, 최근 새로운 치료 컨셉으로 진행되고 있는 종양을 포함한 주변의 다양한 종류의 세포들과 물리적 환경요소들을 아우르는 “종양 미세환경 (tumor microenvironment)”을 대상으로 연구한 논문입니다. 간암의 종양 미세환경에서 방사선에 의하여 CXCL12 유전자의 프로모터 부위에서 후성유전적 변화가 발생하였고, 이로 인하여 이 유전자의 발현이 증가하고 결과적으로 방사선 치료를 방해할 가능성이 있습니다. 이 문제를 해결하기 위하여, 이 후성유전적 변이를 방해하는 억제제 IOX1을 방사선 간암 치료에 함께 이용하는 경우, 종양의 공격성을 완화시켜 악성화를 방지하고 동시에 방사선 치료 효과가 높아질 가능성을 제시하였습니다. 따라서 방사선에 의하여 초래되는 후성유전적 변이를 조절하는 전략은 기초의생명과학자 뿐 만 아니라 방사선 임상 의학연구자들에게 소개 및 도움이 될 만한 좋은 정보라 생각합니다.
- 덧글달기








편집위원
CXCL12 유전자의 후성유전학적 변화 원인을 다양하게 연구중이지만, 방사선에 의해 조절되는 직접적인 원인을 간암세포 모델에서, 후성유전학적 분자기전을 통해 분석한 연구결과라서 흥미로움.
2019-08-30 10:06:30