글로벌 연구동향
핵의학
- [Radiology .] Direct and Indirect Chimeric Antigen Receptor T-Cell Imaging with PET/MRI in a Tumor Xenograft Model종양 이식 모델에서 PET/MRI를 이용한 직접 및 간접 CAR T 세포 이미징
울산의대 / 김석영, 류진숙*
- 출처
- Radiology .
- 등재일
- 2024 Feb
- 저널이슈번호
- 310(2):e231406. doi: 10.1148/radiol.231406.
- 내용
-
Abstract
Background Chimeric antigen receptor (CAR) T cells are a promising cancer therapy; however, reliable and repeatable methods for tracking and monitoring CAR T cells in vivo remain underexplored. Purpose To investigate direct and indirect imaging strategies for tracking the biodistribution of CAR T cells and monitoring their therapeutic effect in target tumors. Materials and Methods CAR T cells co-expressing a tumor-targeting gene (anti-CD19 CAR) and a human somatostatin receptor subtype 2 (hSSTr2) reporter gene were generated from human peripheral blood mononuclear cells. After direct labeling with zirconium 89 (89Zr)-p-isothiocyanatobenzyl-desferrioxamine (DFO), CAR T cells were intravenously injected into immunodeficient mice with a CD19-positive and CD19-negative human tumor xenograft on the left and right flank, respectively. PET/MRI was used for direct in vivo imaging of 89Zr-DFO-labeled CAR T cells on days 0, 1, 3, and 7 and for indirect cell imaging with the radiolabeled somatostatin receptor-targeted ligand gallium 68 (68Ga)-DOTA-Tyr3-octreotide (DOTATOC) on days 6, 9, and 13. On day 13, mice were euthanized, and tissues and tumors were excised. Results The 89Zr-DFO-labeled CAR T cells were observed on PET/MRI scans in the liver and lungs of mice (n = 4) at all time points assessed. However, they were not visualized in CD19-positive or CD19-negative tumors, even on day 7. Serial 68Ga-DOTATOC PET/MRI showed CAR T cell accumulation in CD19-positive tumors but not in CD19-negative tumors from days 6 to 13. Notably, 68Ga-DOTATOC accumulation in CD19-positive tumors was highest on day 9 (mean percentage injected dose [%ID], 3.7% ± 1.0 [SD]) and decreased on day 13 (mean %ID, 2.6% ± 0.7) in parallel with a decrease in tumor volume (day 9: mean, 195 mm3 ± 27; day 13: mean, 127 mm3 ± 43) in the group with tumor growth inhibition. Enhanced immunohistochemistry staining of cluster of differentiation 3 (CD3) and hSSTr2 was also observed in excised CD19-positive tumor tissues. Conclusion Direct and indirect cell imaging with PET/MRI enabled in vivo tracking and monitoring of CAR T cells in an animal model.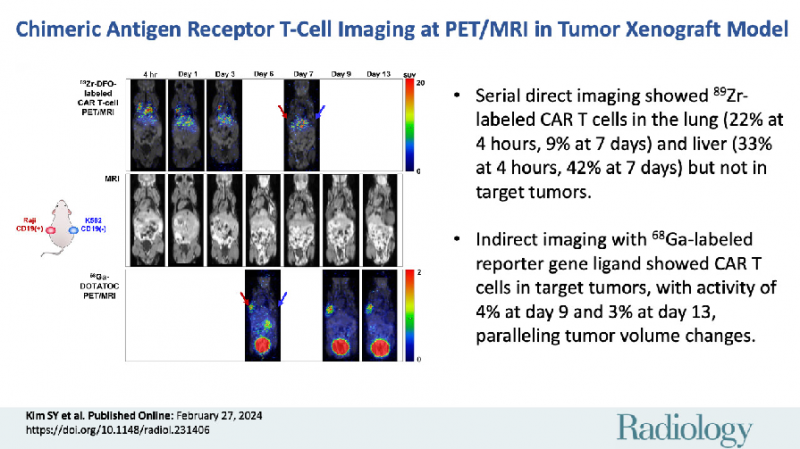
Affiliation
Seog-Young Kim 1, Hyunsu Soh 1, Jin Hwa Jung 1, Eun Hye Cho 1, Hyori Kim 1, Ji-Min Ju 1, Joong Hyuk Sheen 1, Sang Ju Lee 1, Seung Jun Oh 1, Sang-Jin Lee 1, Junho Chung 1, Jin-Sook Ryu 1
1From the Convergence Medicine Research Center (S.Y.K., H.S., J.H.J., H.K.) and Department of Nuclear Medicine (E.H.C., Sang Ju Lee, S.J.O., J.S.R.), Asan Medical Center, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Republic of Korea; Research Institute, National Cancer Center, Gyeonggi-do, Republic of Korea (J.M.J., J.H.S., Sang-Jin Lee); and Department of Biomedical Sciences, Seoul National University, Seoul, Republic of Korea (J.C.).
- 연구소개
- 저자들은 PET/MRI를 사용하여 종양 이종이식모델에서 9Zr-DFO로 직접표지된 세포 영상과 68Ga로 표지된 리포터 유전자 타겟 리간드인 68Ga-DOTATOC를 이용하는 간접 세포 영상을 같이 활용하여 CAR T 세포들의 생체 내 분포를 추적하고 모니터링하는 방법을 연구하였습니다. 연구 결과, 주사 후 초기의 89Zr-DFO 표지된 세포 영상들은 CAR T 세포들의 폐와 간에 축적되는 것을 보여주었으나, 종양에 대한 섭취는 확인할 수 없었습니다. 반면, 동시에 시행된 68Ga-DOTATOC 영상은 타겟 종양에서 증식된 CAR T 세포들의 축적을 보여주었고 이는 종양 크기의 변화와 일치했습니다. 또한, 제거한 종양의 면역조직화학 염색을 통해 리포터 유전자 발현 CAR T 세포들의 축적을 입증하였습니다. 이 연구는 소동물 종양 모델에서 PET/MRI를 사용하여 CAR T 세포의 생체 내 추적 및 모니터링이 직접 및 간접 세포 이미징을 통해 가능함을 확인하였습니다. 향후 다양한 난치성 종양에 대한 CAR T 세포 치료의 개발에서 이와 같은 리포터 유전자를 도입하고 세포 영상들을 활용하면, CAR T세포의 최적화 및 효과성과 안전성 향상에 기여할 수 있을 것으로 기대합니다.
- 덧글달기
- 이전글 [Clin Nucl Med .] Diagnostic Performance of Perfusion-Only SPECT/CT for Chronic Thromboembolic Pulmonary Hypertension in Comparison With Ventilation-Perfusion Planar, SPECT, and SPECT/CT Imaging
- 다음글 [Clin Nucl Med .] 11 C-Acetate PET/CT for Reactive Astrogliosis Outperforms 11 C-Methionine PET/CT in Glioma Classification and Survival Prediction








편집위원
CAR-T 세포는 다양한 악성종양의 치료에 효과를 나타낼수 있는 세포치료제이며 세포치료제가 생체내에서 표적이 되는 종양에 축적여부 확인은 해당세포치료제의 효과를 예측하는데 매우 중요한 요소임. 해당연구은 실험동물 연구를 통해 직접 및 간접리포터유전자 영상기법을 이용하여 생체내 CAR-T 세포의 PET 영상화를 보여준 매우 우수한 연구임. 분자영상 및 핵의학 영상 관련 임상가 및 세포치료술에 관심을 가진 연구자에게 흥미를 끌 것 기초 연구로 생각됨.
2024-03-29 15:25:09